I’m a little behind on my ‘interesting papers’ at the moment, but when this story hit the headlines this morning I wanted to talk further about the concepts[1,2]. The RSC was ahead of the curve with their chemical science article last year[3].
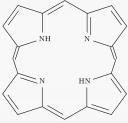
The background is that Lance Twyman, (University of Sheffield) has developed a polymer-porphyrin system with potential for acting as synthetic hemoglobin[4,5]. Hemoglobin is the oxygen carrying protein in blood and has a porphyrin unit. Porphyrin with Fe (II) can bind oxygen reversibly. The protein structure of hemoglobin protects the oxygen at the active site, isolating the porphyrin and protecting against deactivation. Synthetic molecules that mimic the properties of hemoglobin must have a mechanism to protect the oxygen species whilst retaining reversible binding. Twyman uses a hyperbranced polymer based on 3,5-diacetoxybenzoic acid, starting with tetrakis(4-acetoxyphenyl)porphyrin as the core. The advantage of this system over (for example) a dendritic system with similar structure is that the polymer can be made in one step, eliminating the growth-activation strategy that normally plagues dendrimer chemists. There are several examples of dendritic hemoglobin mimics but none have as elegant a synthetic method as this one.
The polymer branch, and the porphyrin core.
Blood is made of many different components, of which hemoglobin is just one. Artificial blood must perform a variety of functions including carrying oxygen around the body, but critically the viscosity of blood must also be maintained. Ideally replacing lost blood should be done with a liquid of similar viscosity to maintain correct cardiac function. One reason this polymeric porphyrin may be additionally useful is that the polymeric component may be sufficient to increase the viscosity of any solution.
The drawbacks of such a system are touched upon briefly by Twyman in the Guardian article [2]. Twyman believes that the polymer will be ignored by the body’s immune system but the experiments to date have been in vitro. Any polymeric material for human use must meet stringent criteria. The polymer and any potential breakdown materials must not be toxic. The polymer can be biocompatible, bioinert, biodegradeable, but close attention must be paid to the ultimate fate of the polymer within the body. Which organs would it accumulate in? How and where and when will it breakdown? These are all questions that must be answered before this system can be applied in medicine, but don’t detract in anyway from the potential life saving usefulness of this discovery.
My only concern regarding the articles I’ve seen to day is the use of the term ‘plastic blood’. I appreciate that it is important to convey concepts to the public but do we really want people associating grocery bags and non-biodegradable landfill disasters with a blood substitute? I think polymer is a better, although less understood term, in this application. Plastic is a little too simplified.
[1] http://news.bbc.co.uk/2/hi/uk_news/england/north_yorkshire/6645923.stm
[2] http://technology.guardian.co.uk/weekly/story/0,,2075544,00.html
[3] http://www.rsc.org/Publishing/ChemScience/Volume/2006/05/artificial_blood.asp
[4] http://www.rsc.org/delivery/_ArticleLinking/DisplayArticleForFree.cfm?doi=b600831n&JournalCode=CC
[5] http://www.shef.ac.uk/chemistry/staff/profiles/twyman.html
Vanillin


![[Contents scheme]](https://i0.wp.com/journals.iucr.org/e/issues/2006/04/00/cf2017/cf2017contents.gif)